For many growing businesses, intellectual property is one of the most important sources of commercial value. It can protect technical differentiation, support brand strength, improve investor confidence and create leverage in licensing, partnership or exit discussions.
Yet, because IP is intangible and frequently misunderstood, it is often managed reactively. Innovations may be discussed publicly or commercialised before protection has been considered. Trade mark gaps may only become apparent once a competitor enters the market. Key contracts with employees, contractors, suppliers or collaborators may be missing, outdated, or signed without properly addressing IP ownership, access rights, and risk allocation.
To bridge this gap and align your commercial goals with legal protection, businesses must take a proactive approach. So, what is an IP audit, and what is the importance of an IP audit for companies looking to scale?
What is an IP audit?
At its core, an IP audit is a comprehensive, systematic review of a company’s intellectual property assets and how intellectual property is managed. It goes beyond a simple administrative list of registered rights. An effective audit assesses the legal validity, commercial alignment, and potential risks associated with your proprietary technology, brands, designs, and trade secrets.
A thorough assessment typically addresses:
- Registered IP: Patents, registered trade marks, and registered design rights.
- Unregistered IP: Copyright, database rights, trade secrets, unregistered designs, open source software use, and know-how.
- Third-party relationships: Freelancer agreements, supplier agreements, and joint-ventures and collaborations that affect ownership.
- Internal IP governance: Founder agreements, employment contracts, IP decision-making processes, and employee training.
Why is an IP audit important?
An IP audit is not just a case of housekeeping; it’s vital for risk management and value creation. The importance of an IP audit can be broken down into two main functions: a snapshot of your current IP position, and mapping out the steps to secure and enhance your IP strategy for the future.
Identifying and cataloguing existing assets
Many businesses do not realise they possess valuable IP until a formal audit brings it to light. For example, a specialised software script, a proprietary manufacturing process, or a proprietary formulation may contain highly protectable innovations. An audit helps you systematically identify and assess these assets, decide how they should be protected, and understand how they may support commercial value
Spotting the ownership and application gaps
Crucially, a robust audit uncovers what you don’t securely own or protect. This is where businesses face the highest risk. A successful audit can reveal gaps such as:
- Missing registrations: Identifying core brand names, logos, or technical inventions that have been deployed commercially but lack formal trade mark or patent filings.
- Ownership vulnerabilities: Exposing weaknesses where contractors, freelancers, or joint-venture partners have contributed to your product development without explicitly assigning the IP rights over to your company.
- Freedom to operate (FTO) risks: Flagging products, services, markets or technical areas where further freedom-to-operate analysis may be needed, and identifying practical steps to reduce or manage third-party IP risk.
How do you conduct an IP audit?
Conducting an IP audit takes your business from a purely compliance-focused position to a commercial, growth-focused one. But if you’ve not engaged with the process before, you may not know where to start.
If you want your audit carried out to the highest possible standards, our experts can help. We work with businesses of all sizes to identify intangible assets and map out a prioritised IP strategy to help you grow.
As a general overview, the IP audit process looks like this:
Step 1 – Scope and Objectives: Start by defining what the audit aims to achieve. Is it preparing the company for an impending Series A funding round, evaluating a brand expansion into new geographical markets, or assessing a newly acquired technical asset?
Step 2 – Information Gathering: Collate internal information, including product plans, marketing collateral, trade secret records, and key IP agreements. We use digital tools to simplify this process for you.
Step 3 – Legal & Commercial Analysis: Evaluate whether your IP is adequately protected. Are your trade marks registered and in the correct classes? Is there technical evidence, such as comparative testing, prototype results, or performance data, that supports the claimed technical advantage of the invention? Are your trade secrets protected by appropriate confidentiality agreements and access controls?
Step 4 – The Audit Report: A well-run IP audit should produce more than a list of registered rights. It should result in a clear, prioritised report identifying the company’s key IP assets, ownership position, protection gaps, third-party risks, contract issues, confidentiality controls and recommended next steps. The most valuable output is usually a ranked action plan showing what should be fixed immediately, what should be monitored, and where future budget should be allocated.
Once these steps have been carried out, your IP audit should act as a useful reference that the business can maintain as new IP is created or acquired.
Your IP audit checklist
If you’re new to the IP auditing process, here’s a checklist that will help steer your thinking in the early stages of identifying, categorising, and managing your commercial IP:
- Inventions & Patents: Have any new technical features, codebases, or formulations been publicly disclosed (e.g., via sales, marketing, or academic papers) before making an informed decision on IP protection.
- Brands & Trade Marks: Are all current brand names, sub-brands, and core logos protected by active trade mark registrations in all your primary jurisdictions and target markets?
- Contractor & Employee Alignment: Do your employment contracts and third-party freelance agreements contain explicit, legally binding IP assignment clauses?
- Trade Secrets & Know-How: Is access to critical proprietary code, chemical formulations, or client databases restricted via technical access controls and comprehensive internal confidentiality policies?
- Third-Party Risk: What steps, if any, have you taken to reduce the risk of your current products or services conflicting with third-party patents or trade marks?
Align your IP strategy with business growth
An IP audit shouldn’t exist in isolation. But for growing businesses, it’s not feasible to recruit a full in-house IP team.
We’re here to help. Our IP specialists can help you to identify your intangible assets, develop a sustainable strategy to gain market traction, and understand your current IP value to set a benchmark from which to grow.
If you want a clearer view of what IP your business owns, where the risks sit, and which actions should be prioritised, our team can help you carry out a practical, commercially focused IP audit.
World Environment Day 2026 is upon us, with its focus this year being on the direct action needed to tackle climate change. A cornerstone of this fight is the expansion of electrification using renewable energy, which comes hand-in-hand with battery developments capable of supporting this expansion.
For many applications, including the much-publicised electric vehicles market, Li-ion batteries are the prevailing energy storage means due to their established efficacy, excellent energy density and cycle life. However, as the electrical revolution progresses, innovation in energy storage solutions is being driven by the host of new applications and situations which electrical implementation must now account for. For instance, sodium-ion batteries have the potential to perform better than Li-ion batteries under lower temperature conditions, avoiding the significant drops in capacity and charging/discharging issues at lower temperatures (e.g. below 0 °C).
Clearly, batteries are an increasingly fundamental technology underpinning most, if not all, aspects of modern life; that is why investment and innovation is of great importance. And there are signs that advanced battery technologies are starting to make the leap from the lab onto the road. Earlier this year, the first passenger vehicle to be mass-produced with a sodium-ion battery was announced. An electric vehicle using a semi-solid-state battery (containing an electrolyte composition that is 95% solid) has recently been unveiled to be coming to the UK in late 2026.
As you might expect from a field with such significance to so many aspects of everyday life, there is a tremendous amount of activity on the patent front. In addition to being essential tools for protecting and commercialising your intellectual property, patents can also provide insights into the technological landscape of a given field. In this article, we’ll take a look at what the patent filing data for a selection of key cell chemistries says about the battery sphere today, and what it might suggest for the future.
Lithium-ion batteries
First released in 1991, the Li-ion battery has, with the help of some further significant developments, exploded in popularity (and only occasionally in aeroplanes) to become the most widespread of modern batteries due to their high energy density, efficiency and long lifecycle. Li-ion batteries have found utility across the spectrum of possible applications, from personal electronics to major components in electric vehicles (EVs). There are a number of cell chemistries that fall within the general category of “Li-ion battery”, varying most significantly in their cathode materials. These include: lithium nickel manganese cobalt oxide (NMC), lithium nickel cobalt aluminium oxide (NCA), lithium iron phosphate (LFP) and lithium cobalt oxide (LCO) cells.
Accompanying this boom in Li-ion battery adoption is a corresponding growth in patent applications in the field.
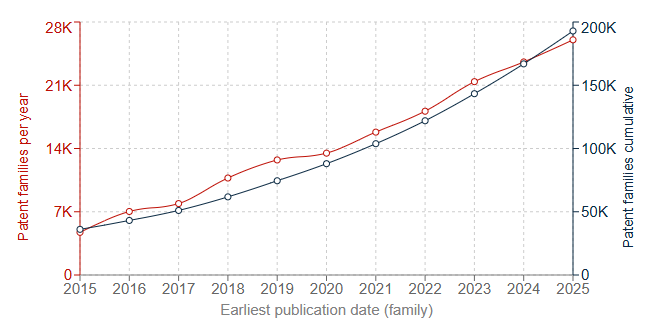
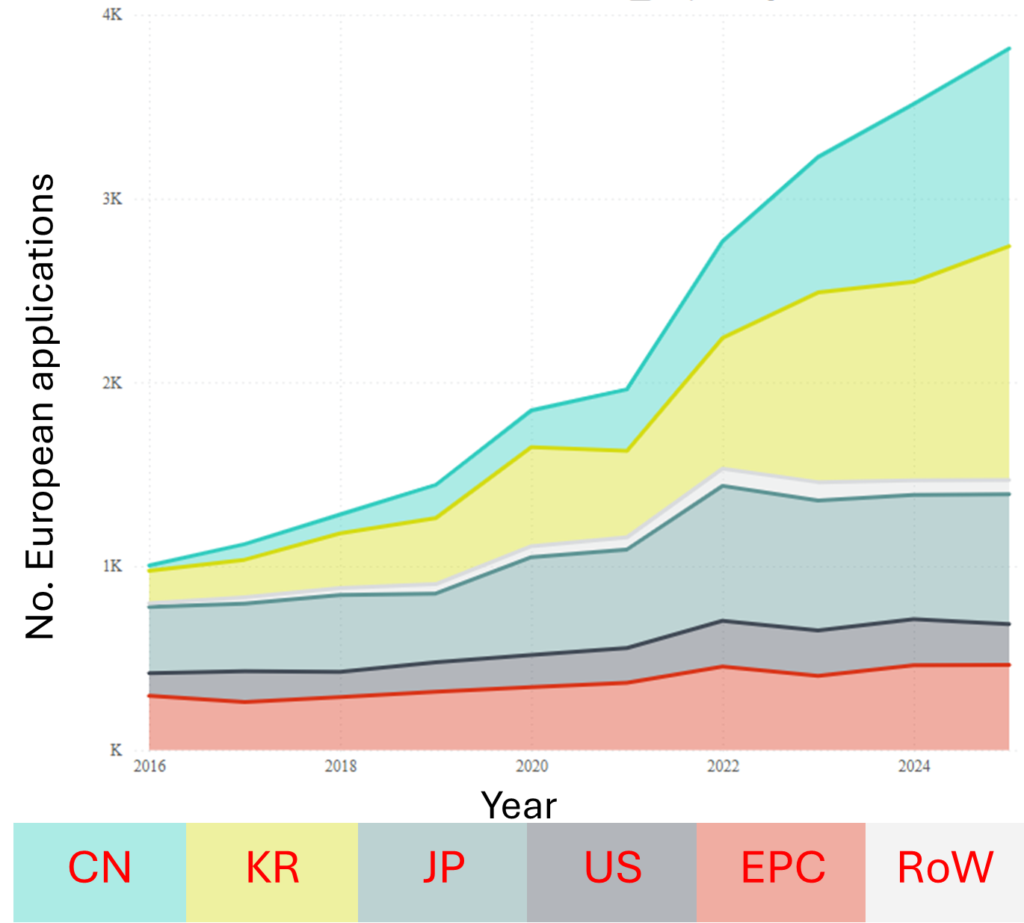
Fig. 1A shows the number of patent families published per year worldwide between 2015 and 2025 relating to Li-ion batteries. Fig. 1B shows the number of EP patent applications published between the years of 2016 and 2025 relating to Li-ion batteries, and includes a breakdown of these according to the applicant’s country.
As can be seen from Fig. 1A, the number of worldwide patent filings related to Li-ion batteries has grown steadily in the years between 2015 and 2025, reflecting their widespread adoption and development: in 2015, around 6000 patent families were published, contributing to a total cumulative number of around 40,000 patent families directed to this subject-matter. By 2025, this had increased to around 26,000 families published that year alone, and a cumulative number of patent families approaching 200,000.
Similar increases are apparent in EP applications, with a particularly notable rise in applications coming from Chinese and South Korean applicants in the past 5 years. Even though Li-ion battery technology, at a base level, has been around for quite some time, patent filings are clearly still growing, highlighting the keen interest and range of developments in the field.
However, one area of Li-ion battery technology that may still be in its (relative) infancy is recycling. In addition to the clear environmental benefits of recycling, Li-ion batteries contain several valuable elements, the efficient recovery of which is desirable from an economic standpoint. Given the typical lifespan of 10 to 20 years of Li-ion batteries and the increasing number reaching end-of-life stages in the near future, there may be a growing incentive to implement effective and economical recycling methods. The development of economical methods may be especially important for certain cell chemistries (such as LFP cells) which present more of an economic challenge due to the lower content of the more valuable elements, such as cobalt. While the number of patent applications directed towards such recycling methods seems to be growing year-on-year, it still remains a relatively minor portion of overall filings, with approximately 400 patent families published in 2025.
Sodium-ion batteries
Sodium-ion batteries are an emerging technology with significant potential for further innovation and applicability to current battery implementations. Whereas Li-ion batteries rely on lithium ions as charge carriers, sodium-ion batteries utilise (unsurprisingly) sodium ions. Otherwise, at least in their current forms, sodium-ion battery architecture is broadly similar to Li-ion battery cells, although the use of sodium ions allows for different electrode materials that often involve less expensive elements. For instance, current sodium-ion cells often employ layered transition metal oxide or Prussian white cathodes. Further cost benefits arise due to the abundance of sodium, as compared to lithium, as a key component, which can help to alleviate possible supply chain issues.
As already mentioned, sodium batteries also come with certain performance advantages over Li-ion batteries, albeit (at least at present) mainly only under specific conditions. In particular, sodium batteries can function effectively over a wider range of temperatures while maintaining good efficiency and capacity metrics that can be roughly comparable to Li-ion batteries under normal conditions.
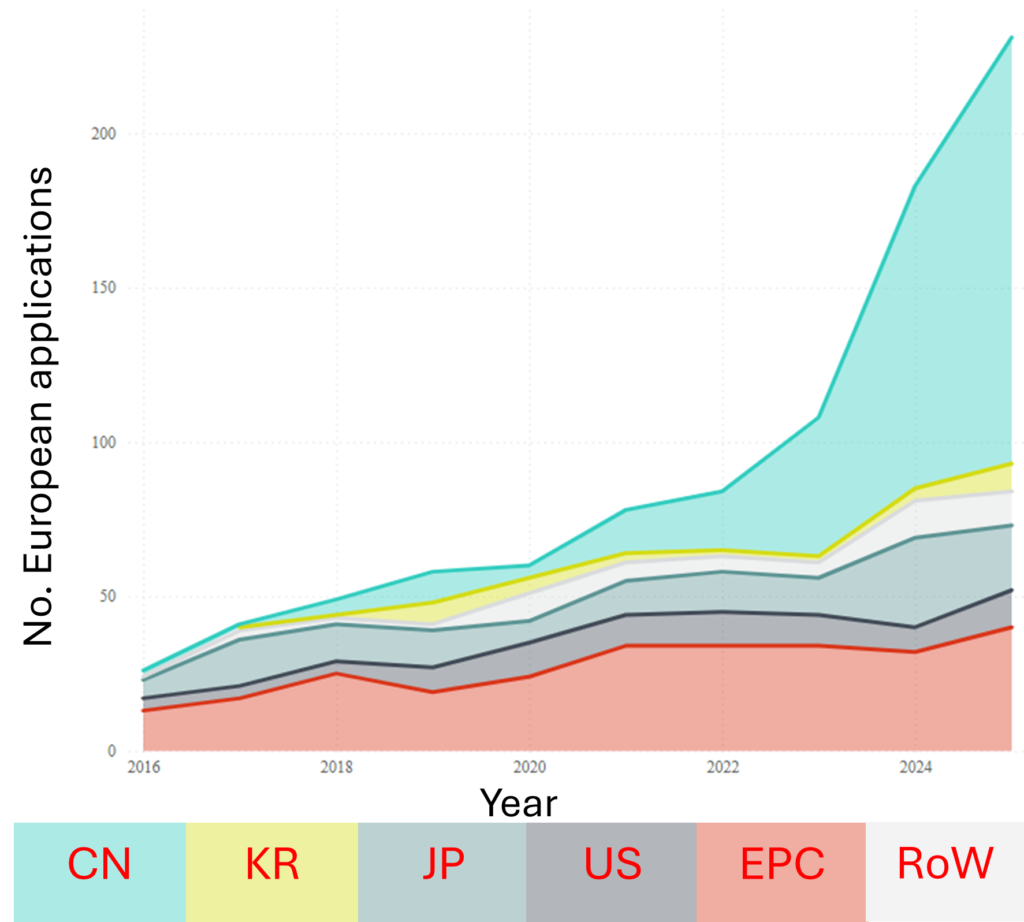
Although initial research into sodium-ion batteries roughly coincided with Li-ion batteries, the early promise of Li-ion cells caused their sodium-based sibling to fall by the wayside. However, commercial interest began to be renewed in the 2010s; this is reflected in the patent filings, as shown in Figs. 2A and 2B.
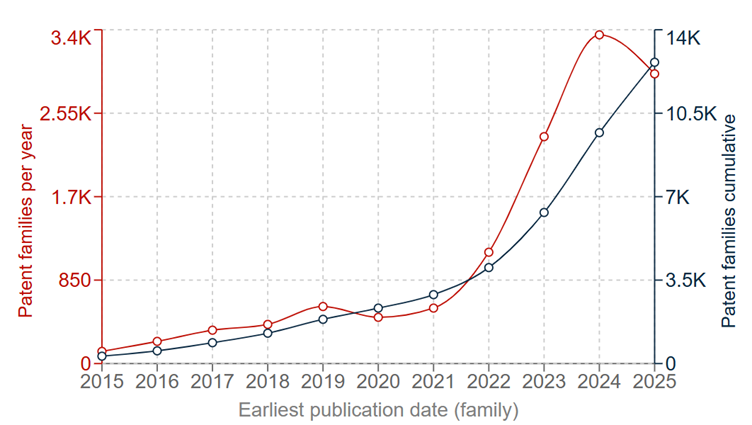
From only a handful of patent families published per year in the mid 2010s, patent filings grew steadily to about 500 patent families in 2021. From this point, there has been a surge in applications, with nearly 3,500 patent families filed in 2024 alone, adding to a cumulative total of around 10,000 families. It remains to be seen, however, whether the slight decline in 2025 represents a genuine cooling in interest, or simply a minor fluctuation.
Certainly, the EP filings show no decline in patenting activity, which is being driven primarily by China who are a clear frontrunner in sodium-ion cell chemistry. However, the sodium-ion battery field is one of the relatively rare situations where EPC countries are in a comparatively prominent position (in terms of filing numbers).
Comparing these numbers with the Li-ion data, patent filings for sodium-ion batteries are only at a similar level to Li-ion batteries in the early 2010s. This could point to there being plenty of potential for further innovation and development, particularly if their commercialisation is successful.
Lithium-sulfur batteries
Lithium-sulfur (LiS) batteries are developments of more typical Li-ion batteries, based on replacing the cobalt or iron-based cathode material with one consisting of S8 sulfur. In these batteries, lithium ions are stored as lithium sulfide (Li2S) during discharge, creating the potential for a cell with much higher specific energy than allowed for by the intercalation of Li-ions in conventional Li-ion cells.
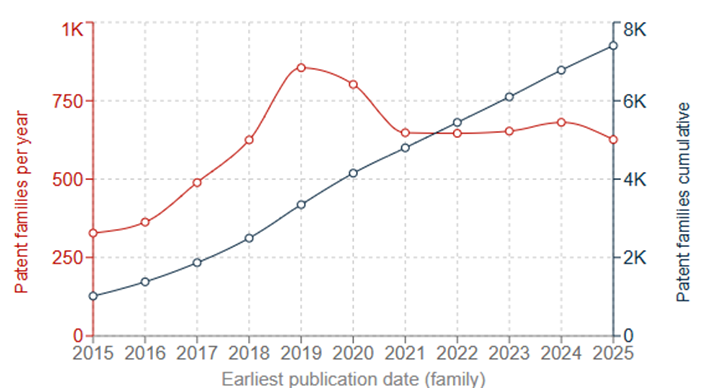
While the potential benefits from developing a successful LiS battery could be enormous, there are still barriers to overcome in the technology if they are to be successfully commercialised, such as electrode expansion and polysulfide shuttling. These barriers may be posing a challenge, or at least acting as a deterrent, since LiS battery technology appears to remain in its very early stages compared to conventional Li-ion batteries, despite the first LiS prototype batteries having been around since as early as the 1960s. This is reflected in the worldwide patent filings in Fig. 3A: from about 300 patent families published in 2015 of a cumulative 1,000 patent families, filings reached a peak of about 850 families in 2019 but have since diminished to a consistent number of about 600 patent families filed per year. These numbers are similar to those of Li-ion batteries over 20 years ago.
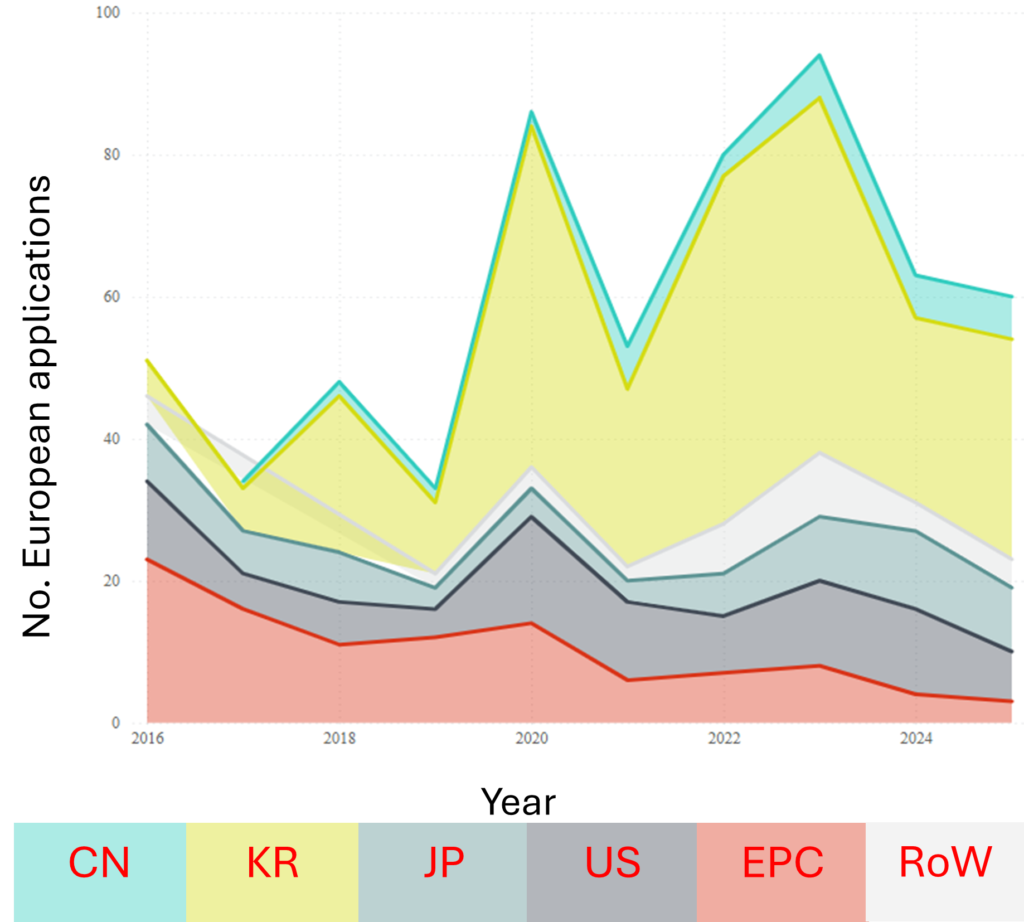
A lack of widespread adoption is also suggested by the EP data in Fig. 3B, which shows both a similar trend of declining applications in the past couple of years, and an overwhelming dominance in the field by South Korean applicants.
Solid state batteries
Conventional Li-ion batteries typically make use of liquid electrolytes, consisting of lithium salts in various organic solvents. However, solid state batteries aim to make use of a solid state electrolyte for conducting the charge carrying ions between the electrodes, which could provide significant improvements in terms of energy density and safety. Possible solid state materials naturally require high ionic conductivities, and a large variety of material classes to meet this requirement have been, and are currently being, explored. These include: polymer electrolytes; inorganic electrolytes such as oxide solid and sulfide-based electrolytes; and composite electrolytes that aim to incorporate aspects of both of these.
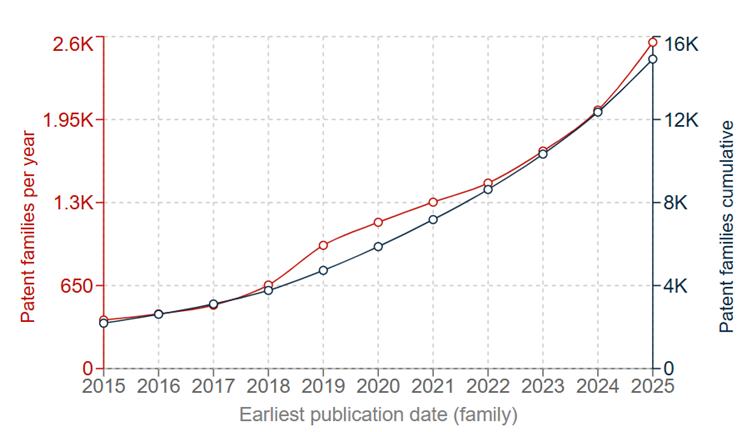
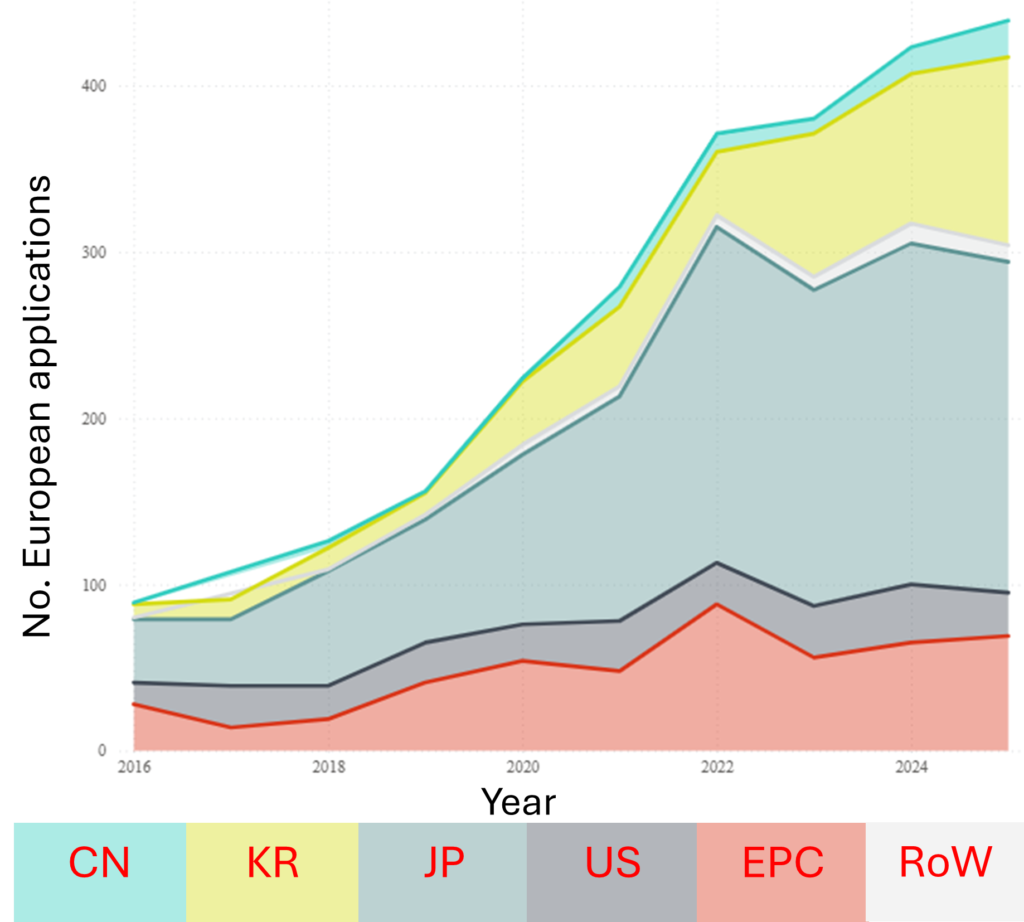
Growing demand for compact, safe, and high-capacity batteries across EVs and consumer electronics has seen solid state battery development start to approach manufacturing and early commercialisation. Correspondingly, the overall trend – both worldwide and at the EPO – is that of significant increases in filings. Although the technology is nowhere near as mature as general Li-ion batteries, and filings are currently only at a similar level to Li-ion batteries 15 years ago, patenting activity in solid state batteries is only likely to increase in the coming years.
Batteries and the clean energy transition
A crucial step to tackling climate change is moving away from fossil fuels and transitioning to net zero. To achieve this goal, we need to make a marked shift towards cleaner energy sources, such as solar and wind. However, there are obstacles which need to be overcome to ensure that these sources can provide sufficient power to fuel our ever-growing demand. Solar panels only generate electricity when the sun is shining. Wind turbines only generate electricity when the wind speeds are suitable. Therefore, this electricity must be stored and later released when supply falls or demand rises.
Innovation in battery technology is helping to solve this major challenge in the clean energy transition, providing a way to store energy and facilitating the widespread adoption of low-carbon electricity systems. Batteries are also essential in the electrification of transport which is another important measure for reducing greenhouse gas emissions.
Lithium-ion batteries currently play a major role in both electric vehicles and energy storage systems. Their relatively high energy density allows large amounts of energy to be stored in a compact space. On the other hand, sodium-ion batteries could be emerging as the holy grail of energy grids due to the greater availability of sodium. Although they store less energy per kilogram, this may not be of consequence for the storage of renewable electricity, and their relatively lower cost and reduced reliance on critical minerals could accelerate the clean energy transition. In terms of the electrification of vehicles, solid-state batteries may take us one step further. Whilst they are largely in a pre-commercial stage, it is suggested that they can offer a higher energy density, faster charging, improved safety and longer battery lifetimes. This could be revolutionary for vehicles like long-range passenger transport, heavy goods transport, or even aviatic and marine vehicles.
Patent strategy
Of course, there are far more possible battery technologies in development, and even more avenues for development, than have been discussed here. There undoubtedly remains a wealth of opportunity, across the entire field of battery development, for the discovery of the next transformative breakthrough.
However, as the data above indicates, there exists an extensive minefield of patent disclosures and patent rights. Is important to note that, despite the abundant prior art these patents represent, disclosures of broader subject-matter are not necessarily a bar to obtaining patent protection for further optimisations of that subject-matter. However, questions around freedom-to-operate would likely need to be answered. In such a busy field, navigating this maze of prior art and prior rights requires careful consideration and a comprehensive IP strategy.
Maintaining our Gold Tier ranking, Mathys & Squire is delighted to be recommended in the 2026 edition of IAM Patent 1000: The World’s Leading Patent Professionals.
IAM is widely regarded as a leading authority in recognising excellent private practice patent expertise. Its rankings highlight top firms and practitioners through detailed qualitative research, considering elements such as previous work, technical capabilities and market presence.
We have been recognised as “a professional and well-respected firm, delivering high-quality legal work with a strong emphasis on responsiveness and efficiency.” Additionally, our team, “provides clear, practical advice and demonstrates a well-coordinated approach across matters. Overall, the experience of working with them is consistently positive thanks to their reliable and value-driven service.”
This year we have been ranked Gold Tier for prosecution in the United Kingdom: England and Wales, and have also been awarded the Silver Tier for the European Patent Office, highlighting the consistent, high quality service we provide for our clients.
Recommended individuals
In addition to our firm ranking, our Mathys & Squire attorneys have been recognised as Recommended Individuals.
Life Sciences & Chemistry team
- Chris Hamer is “attentive and brings a creative, well-informed approach to developing and protecting patent portfolios. He provides clients with practical advice and strong commercial insight, helping them define the right IP position and align it with investor expectations. He also supports assurance and due diligence activities, including freedom-to-operate analyses, and contributes to shaping overall business and portfolio strategy. His input plays a central role in strengthening companies’ development and long-term resilience”.
- Martin MacLean “works closely with scientists to review inventions, identify key technical details, and deliver high-quality patent applications. He supports drafting, prosecution, investor due diligence, and portfolio development. Providing pragmatic, strategically grounded advice, he combines credibility and professionalism with a clear, no-nonsense approach. From early-stage support through to core patent families, Martin helps underpin long-term success, with a strong ability to identify both technical significance and commercial potential”.
- “Michael Stott handles a wide-ranging practice spanning pharmaceuticals, materials science and polymer technologies, including responsibility for a significant global portfolio for Stratasys Inc. relating to 3D-printed materials.”
- Philippa Griffin “provides expert support in European patent prosecution and opposition proceedings, handling complex scientific subject matter with clarity and precision. She drafts and amends claims with strong legal and technical insight and represents clients with professionalism and composure. With a deep understanding of scientific and legal frameworks, Philippa delivers robust, well-reasoned filings. Her advocacy is strategic and credible, and her clear, collaborative communication supports the effective handling of complex matters.”
- “Craig Titmus supports clients through patent approvals and subsequent grants, delivering work that is both effective and commercially balanced. He demonstrates a strong ability to align costs with value, providing practical guidance throughout the process. Craig communicates complex IP concepts in a clear and accessible way, making them understandable for non-technical stakeholders and supporting informed decision-making.”
- “Anna Gregson works on complex biochemistry patent applications, including coordinating the co-filing of multiple related applications. She is diligent and detail-oriented, delivering high-quality work with consistency. Anna responds effectively to tight timelines, maintaining strong standards of service and reliability throughout.”
- “Stephen Garner is a trusted adviser to global pharmaceutical companies, handling the drafting, prosecution and opposition of patent portfolios, including the coordination of international strategies and supplementary protection matters, ensuring that clients’ key assets are both protected and effectively managed across jurisdictions.”
IT & Engineering team
- “Dani Kramer is a key figure in computing and software, advising on large-scale European portfolios and managing complex prosecution work, including technically demanding EPO examination and appeal proceedings across high-performance computing, semiconductors and networking technologies.”
- “Telecoms expertise is anchored by Alan MacDougall, who advises on the protection and defence of patent portfolios, including opposition work before the EPO, with a focus on preserving competitive advantage in fast-moving markets. “
- “James Pitchford strengthens the physical sciences offering through close collaboration with inventors and in-house teams, overseeing the drafting of original applications and coordinating international filing strategies to secure protection for technically sophisticated innovations.”
- “Andrew White brings cross-sector experience spanning medtech, software, telecoms and automotive technologies, with particular strength in deep tech. He has worked closely with Modini Limited to develop and prosecute a patent portfolio for its UAV technologies, covering areas such as avionics, engines and launch systems.”
- “Nicholas Fox offers a rare blend of legal and technical expertise as both solicitor and patent attorney, advising on UPC strategy and aligning opposition approaches across EPO and UPC proceedings to ensure consistency in multi-forum disputes.”
Managing Associates Alex Elder and Matthew Morton (Life Sciences & Chemistry) are also featured as Recommended Individuals.
You can see our ranking on the IAM website here.
We are delighted to announce that two of our Partners in our London office have been promoted.
Dani Kramer has been promoted to Senior Equity Partner and joins the Senior Equity Management Team, whilst Max Thoma has joined the Equity Partnership. These promotions have been made with immediate effect.
These promotions reflect our firm’s commitment to recognising and awarding the talent across our team and the valuable contributions made by its members.
Dani Kramer is appointed Senior Equity Partner
Dani works for a number of large corporations, drafting and prosecuting patent applications, and managing their international patent portfolios. He focuses on the fields of AI and machine learning, microprocessors, communication technologies, internet television, software, and electrical and electronic engineering. Dani is recommended in the latest edition of IAM Patent 1000, the three most recent editions of IAM Strategy 300 and multiple editions of The Legal 500.
Max Thoma is appointed Junior Equity Partner
Max has extensive experience in drafting and prosecuting patent applications, and handling opposition proceedings at the EPO, working across a variety of fields, in particular, computer-implemented (software) inventions and engineering-related fields. He enjoys supporting multinational businesses on major contentious work, as well as working closely with startup businesses through the process of protecting their innovations. He has been featured in multiple editions of Managing IP’s IP STARS directory which recognises IP practitioners.
Partner Martin MacLean, says, “These promotions demonstrate our dedication to career progression here at Mathys & Squire. On behalf of the whole firm, we congratulate Dani and Max on their ongoing hard work and the high-quality service which they deliver to our clients, and are excited to welcome them to the next stage of their time at Mathys & Squire. They will continue to be an immense asset to our company, as it grows and strengthens its leadership team.”
Many growing businesses now recognise the value of an outsourced or fractional General Counsel: senior legal expertise, often with years of practical in-house experience, embedded in the business, but without the cost, risk or long-term commitment of a full-time senior hire. It is a model that gives ambitious companies access to experienced judgement at the point they need it most.
For many SMEs and businesses looking to improve their IP position, there is a strong case for applying the same model to intellectual property.
IP is often central to the value of an innovation-led business. It can protect technical advantage, strengthen brand position, support investment, underpin partnerships, improve negotiating leverage and increase value in a sale or exit. Yet despite its importance, IP is often managed reactively or in a fragmented way, with advice sought only when a problem arises or an opportunity is missed, such as disclosing innovation without protection, encountering a trade mark issue, an ex-employee setting up in competition, investor questions or contract negotiations.
That approach can leave value on the table, create costly delays and expose the business to avoidable risks.
Good IP management is not just about registering rights. It is about making informed, commercial decisions that will shape the future of the business: what to protect, how to protect it, what not to spend money on, how to manage risk, how to structure ownership, how to deal with collaborators and contractors, and how to ensure the IP strategy supports the wider business plan.
The challenge for many SMEs is that they do not need, and often cannot justify, a full-time senior IP hire. But they do need access to experienced IP leadership that can support them through the broad range of real-world IP matters that impact their business now and in the future.
Outsourced IP management helps bridge that gap.
An experienced external IP advisor can provide senior-level strategic, legal and operational support without adding permanent headcount. The support can flex as the business grows: light-touch guidance at an early stage; more regular input during product development, investment rounds or international expansion; and deeper support when preparing for due diligence, licensing, acquisition or exit.
This scalability is one of the model’s key advantages. Businesses can access the right level of expertise at the right time, while maintaining control over cost and avoiding the overhead of building an internal IP function too early.
Our clients work with a dedicated IP advisor who gets to know the business, its technology, its commercial objectives and its key stakeholders. That continuity matters. The best IP advice is rarely given in isolation; it depends on understanding the commercial context, the competitive landscape, the internal priorities and the long-term direction of the company.
At the same time, the dedicated advisor can seamlessly call upon the wider expertise of Mathys and Squire whenever required. That may include specialist patent drafting, trade mark protection, designs, freedom to operate, licensing, due diligence, disputes or valuation support.
For founders and management teams, this provides a practical middle ground: senior IP leadership without senior headcount; strategic continuity without building an internal IP function too early; and access to specialist expertise as needs evolve.
For many SMEs, IP is too important to be left to chance, but a full-time senior hire may not yet be the right answer.
Outsourced IP management offers a scalable, commercial and effective alternative.
Is your IP strategy keeping pace with your business growth?
Our fractional IP advisor service gives you access to senior strategic, legal and operational IP support on a flexible basis. Click here to learn more.
If you are looking for senior IP input but are not ready for a full-time hire, get in touch here.
On 4th May, the UPC revoked the ‘remdesivir’ anti-viral drug unitary patent owned by Chinese research institute AMMS and rejected all limitations proposed by the patentee (case ID UPC_CFI_552/2025).
Granted on 18 June 2025, the patent EP 3854403 claimed the use of remdesivir for the treatment of COVID-19, threatening the commercial operations of Gilead Sciences – the original developers of remdesivir. Gilead brought revocation action on the day of grant on the grounds that the claimed second medical use of remdesivir for treating COVID-19 was not inventive and lacked sufficiency.
The background
Remdesivir is a prodrug that is metabolised within cells after it is administered. Once within cells, it is processed into a nucleotide analogue that mimics adenosine (an RNA building block). This nucleotide analogue halts viral replication of RNA viruses by disabling a key viral enzyme, the RNA polymerase, which is required for copying the viral RNA genome. Prior to 2020, remdesivir had not been clinically proven to treat any viral infections, but preclinical research had shown that the drug appeared to be promising for treating infections by coronaviruses SARS and MERS.
Gilead has patents for the compound and for the use of the compound for treating coronavirus infections, the latter of which was filed in 2016, years before SARS-CoV-2 (SARS2) was identified. However, AMMS managed to file an application for the second-medical use of treating viral infections by the newly identified SARS2 before the pandemic was even declared by WIPO and shortly before clinical trials began in China in early 2020.
Narrow second medical use claims are routinely accepted by the EPO, even if an identical treatment for the broader class of a disease (the “first medical use”) has been disclosed before the priority date of the second medical use patent application. However, the requirement for an inventive step still applies and this can be undermined if the skilled person has been provided with some reasonable expectation of success.
The decision
In the case of EP 3854403, the Court dismissed the claimant’s sufficient objection but revoked the patent on the grounds that the second medical use lacked an inventive step. The decision issued by the Central Milan Court states that the treatment of COVID-19 with remdesivir was an obvious choice for the skilled person at the application date because the viral genome had been made publicly available before the priority date and it was therefore known to the skilled person that SARS2 was highly similar to SARS1.
The Court further noted that experts had speculated publicly that remdesivir would be a good candidate anti-viral treatment due to the positive results from pre-clinical experiments on SARS1 and MERS. The decision highlights how the UPC applies a strict approach to assessing the inventive step of second medical use and indicates that patentees must be wary of any ‘hints’ to the treatment’s effectiveness published prior to the applications priority date.
Gilead is also pursuing an Opposition against the patent at the EPO. The UPC and EPO are distinct entities so the outcome of the opposition could differ from the UPC’s recent decision, but it seems likely that the EPO will take a similar approach since parallel decisions to date have remained broadly uniform. While the reasoning for decisions occasionally differs, the Enlarged Board of Appeal decision G1/24 regarding claim interpretation demonstrated an intent to bring EPO case law in line with UPC rulings.
Battles for pandemic innovation IP
While the commercial significance for these technologies is huge, several pharmaceutical companies publicly pledged not to enforce their patent rights to allow mass production of life saving drugs and vaccines at a time of global crisis. These allowances were short-lived, ceasing in May 2023 when the pandemic was officially declared to be over, and contentious proceedings are ongoing for a number of critical patents.
The disputes have largely been centred around patents for the widely distributed COVID-19 mRNA vaccine filed early in the pandemic. The most high-profile cases revolve around ongoing infringement and invalidity proceedings between Moderna and BioNTech/Pfizer.
In the wake of the pandemic, applications of thousands of patent families directed to Covid-19 related technologies were filed in the space of a few short years. While many patents filed in 2020 following the characterisation and subsequent proliferation of SARS-COV-2 have been granted, many divisional applications and later developments are still pending prosecution, and may yet be the subject of new infringement and revocation proceedings.
The impact of these patent disputes extends further than the ownership of the IP itself; as the decisions handed down by the UPC and European Boards of Appeal shape the case law on inventive step, plausibility and medical use claims.
Users of the European patent system will be familiar with the EPO’s long-standing practice of requiring the description to be brought into line with amended claims, both before grant and in post-grant proceedings. The EPO stands alone among the major global patent offices in having such a requirement, and in recent years both the legal basis for this practice and the extent of amendments required (if any) have become highly controversial.
Oral Proceedings took place today, 8 May, before the EPO’s Enlarged Board of Appeal. The referral asks whether the EPC requires adaptation of the description following claim amendments, and if so, which provision supplies the legal basis. This article summarises the background, the route to the referral, the written submissions, and the main arguments at today’s hearing.
Background: what’s the debate all about?
For many years, the EPO has required applicants, before grant, to bring the description into line with any allowable amended claims. The same requirement arises post-grant where a patent is maintained in amended form. Historically, this was often a relatively light-touch exercise. However, in 2021 the EPO adopted a new policy requiring proprietors either to delete embodiments no longer covered by the amended claims, or to mark them “prominently” as not falling within the claimed invention. This turned what had been a routine procedural step into a major point of controversy.
Objections to this requirement are both legal and practical. Aside from the time and expense involved in making extensive description amendments (particularly in the case of long or complex specifications, or those originally drafted in a non-EPO style), concerns have been raised about interference with claim interpretation during litigation and the risk of introducing added matter. Inconsistent practice between different examining/opposition divisions and Boards has added to the uncertainty.
How did this end up before the Enlarged Board?
Given the controversial change in practice, it was inevitable that users of the EPO began to push back. Starting with decision T 1989/18, a minority line of Technical Board of Appeal decisions began to question whether the EPO’s practice had any legal foundation. This provoked a reaction from other Boards seeking to provide legal justification for the EPO’s practice, such as in T 1024/18 and T 3097/19.
In the case underlying today’s hearing, T 697/22, the Patentee amended the claims during opposition-appeal proceedings. The amended claims were found to meet the requirements of added matter, sufficiency of disclosure, novelty and inventive step. However, the Board found that the claims were inconsistent with the description as amended at first instance. A further amendment to the description was inadmissible for procedural reasons. The question therefore arose as to whether that inconsistency itself prevented maintenance of the patent as amended, whether under the support requirement of Article 84 EPC “or any other requirement of the EPC”.
In view of the divergence in the case law, the Technical Board of Appeal referred the following three questions to the Enlarged Board:
1. If the claims of a European patent are amended during opposition proceedings or opposition-appeal proceedings, and the amendment introduces an inconsistency between the amended claims and the description of the patent, is it necessary, to comply with the requirements of the EPC, to adapt the description to the amended claims so as to remove the inconsistency?
2. If the first question is answered in the affirmative, which requirement(s) of the EPC necessitate(s) such an adaptation?
3. Would the answer to questions 1 and 2 be different if the claims of a European patent application are amended during examination proceedings or examination-appeal proceedings, and the amendment introduces an inconsistency between the amended claims and the description of the patent application?
Written proceedings before the Enlarged Board
In written submissions ahead of the hearing, both the Patentee and the Opponent argued against the EPO’s current practice, though their positions were not completely aligned with one another. The Patentee essentially argued that there was no requirement for adaptation whatsoever. The Opponent took a more nuanced position advocating a significant narrowing of the circumstances in which adaptation of the description could be required, essentially restricting these to situations in which inconsistencies genuinely interfered with the assessment of patentability.
Submissions on behalf of the President of the EPO unsurprisingly defended the EPO’s current practice, primarily on the basis of the “support” requirement of Article 84 EPC and grounds of public policy relating to harmonisation with national courts and the UPC and promotion of “legal certainty for the public as to the scope of the patent monopoly”.
Given the significance of the questions referred, it is not surprising that over 40 amicus curiae briefs were submitted, mainly by patent attorneys, professional associations, and representatives of industry. The vast majority of these argued against any requirement for description amendments, or for a significant restriction of the EPO’s current practice. Common arguments included a lack of clear legal basis, concerns about interference with the competence of national courts and the UPC, the absence of comparable requirements in other major jurisdictions, procedural inefficiency and the risk of unintended consequences such as introduction of added matter.
Looming large over the proceedings was the Enlarged Board’s own recent decision in G 1/24, which held that the description and drawings should always be used to interpret the claims. For the pro-adaptation side of the argument, this meant that inconsistencies now carry greater significance and must be removed. For the anti-adaptation side, G 1/24 instead meant that inconsistencies could be dealt with as an act of interpretation, taking into account the primary role of the claims for determining both patentability and scope of protection.
The Enlarged Board’s preliminary opinion, issued about two months before the hearing, shed relatively little light on its thinking. The Enlarged Board identified two types of inconsistency: those which do not cause noncompliance with the EPC (thereby necessitating no amendments), and those which do (thereby necessitating amendment). However, the Enlarged Board left it open as to how such inconsistencies were to be identified and categorised, or how they might give rise to noncompliance with the EPC.
Oral proceedings before the Enlarged Board
In line with its written submissions, the proprietor argued strongly against any requirement for description amendments. At the heart of its oral submissions was an argument that the “support” requirement of Article 84 EPC merely requires that the claimed subject matter must be derivable from the description, and that amendment of the claims does not impose a reciprocal obligation to amend the description. This argument was based on the plain wording of Article 84 in both English and French, and on a survey of secondary sources including the travaux préparatoires to the EPC.
The proprietor argued that the EPO’s practice rests on a conflation of distinct provisions relating to the role of the claims in defining the subject-matter for which protection is sought on the one hand (Article 84, first sentence) and the requirements of clarity and support on the other hand (Article 84, second sentence). Any inconsistency between the description and claims could be dealt with as a matter of interpretation, recognising that the claims remain controlling for determining the scope of protection (in line with Article 69 EPC) while the description and drawings are consulted to understand their terminology (in line with G 1/24).
The proprietor also emphasised that Article 84 is not a ground for revocation in post-grant proceedings but that a strict adaptation requirement could result in Article 84 becoming a ground of revocation through the back door. Following G 3/14, “pre-existing” clarity or support issues under Article 84 in a granted patent cannot generally be reopened in opposition. A broad requirement for adaptation could therefore also create the odd result that some inconsistencies must be removed while others must be tolerated.
The Opponent took a more nuanced position, but also resisted any broad requirement for conformity between claims and description. It accepted that claim interpretation after G 1/24 is a unitary process in which the description and drawings are consulted together. However, it argued that any real interpretative problem should be addressed as part of the substantive examination of patentability, sufficiency or added matter. Once the claims have been found allowable, there should not be a further, separate exercise of combing through the description for possible inconsistencies, unless a specific inconsistency creates serious doubt about the scope of the claims. The opponent also argued that aggressive description adaptation can distort the balancing exercise between the interests of the patent proprietor and those of third parties as required by Article 69 EPC and its Protocol on Interpretation, tilting the balance too far toward a literal reading of the claims by excising broader subject-matter providing a wider context for their interpretation.
The EPO’s representatives defended the need for adaptation, but in contrast to their written submissions, focused principally on public policy arguments regarding the need to generate certainty for third parties rather than elaborating significantly on the underlying legal basis for current practice. The EPO devoted a significant proportion of its oral submissions to an argument that adaptation of the description would reduce the risk of divergent interpretations of the scope of protection and divergent outcomes in proceedings in different member states. The EPO did not explain how it reconciled this position with its assertion that this did not interfere with the competence of the national courts when interpreting the claims under Article 69 EPC in the context of infringement proceedings.
The Enlarged Board’s questioning was led by the English patents judge Richard Arnold, sitting as an external member of the Board. Questioning focused on several recurring issues:
- First, if G 1/24 requires the description always to be consulted when interpreting the claims, does that support a requirement for adaptation, or does it mean that courts and skilled readers can resolve inconsistencies without amendment – and if so, can such inconsistencies always be resolved as a matter of interpretation?
- Secondly, why should courts be required to deal with sub-optimal, unclear or unamended descriptions if inconsistencies can be resolved by amendment in proceedings before the EPO?
- Thirdly, if claim interpretation is truly a unitary process in which claims and description are to be read together at all stages, is the EPO’s normal practice of deferring description amendment until an allowable set of claims has been identified legally coherent, or should parties logically be required to submit an amended description with each and every amended claim set (a proposition which neither the parties, nor the EPO, nor the Enlarged Board appeared to be willing to endorse)?
Next steps
As is typical of Enlarged Board proceedings, no decision was announced at the conclusion of the hearing. The Enlarged Board’s answers and their reasoning will follow in writing, most likely at some point later this year.
If the written decision follows the contours of the Enlarged Board’s preliminary opinion and the points discussed at the hearing, it seems unlikely that the Enlarged Board will abolish the requirement for description amendments altogether. Equally, however, the maximalist position adopted by some within the EPO under its current practice appeared to be under pressure from the Enlarged Board’s questioning. The main question therefore appears to be how the Enlarged Board will define the threshold differentiating between harmless inconsistencies or those which can be resolved as a pure exercise of claim interpretation, and those which it views as genuinely problematic and necessitating amendment.
Mathys & Squire is proud to share that a selection of our Partners have been featured in the 2026 edition of World Intellectual Property Review (WIPR) Leaders.
Partners Claire Breheny, Anna Gregson, Christopher Hamer, Dani Kramer, Alan MacDougall and Andrew White have all been recommended in this year’s guide.
WIPR Leaders is a reputable point of reference providing an insight into the top intellectual property (IP) practitioners who are spearheading the future of IP across six continents. The entrants are chosen based on highly credible peer-to-peer endorsement, as well as their individual credentials. Inclusion in the guide highlights our attorneys’ innovative approach to IP legal practice, and comprehensive ability to advise clients on complex, interdisciplinary IP matters.
The 2026 guide is available on the WIPR website here.
We extend our congratulations to the attorneys who have earned a place in this year’s ranking.
Partner Rebecca Tew and Managing Associate Adam Gilbertson have been featured in World Intellectual Property Review, Business Insider (UK and Spain) and the Retail Times providing commentary on the rise in trainer patent applications by top-level sports brands.
Their commentary examines how growing attention to the sport, aided by local running communities and large-scale events, has encouraged investment in trainer innovation and increased IP protection, particularly by industry giants Nike and Adidas.
Read the extended press release below.
- Nike had 374 trainer patent applications published in the past year – nearly three times more than Adidas, its nearest competitor*
- Patent activity moves beyond traditional sportswear giants, with brands like On breaking into the top five filers
The number of trainer patent applications by the largest sportswear brands jumped to 791, up 31% from 601 in the previous year*, as companies race to develop new footwear technology, says intellectual property law firm Mathys & Squire.
Mathys & Squire says the rise in patent filings in this field reflects the growing investment in research and development of new technologies and innovations aimed at improving the performance of amateur and recreational runners, who are becoming increasingly competitive.
Specifically, the rise of communities like Strava and Parkrun, adoption of professional training methods, and the increased use of data and performance tracking technologies, has made amateur / recreational runners become more competitive than ever. Recreational runners are also more willing (and able) to take on demanding events, such as the London Marathon and Ultra-Trail du Mont-Blanc.
Sportswear companies are looking to capitalise on this by developing trainer technologies that will help amateur athletes run further, for longer. For example, leading trainer manufacturers are locked in a race to create near-weightless shoes for runners; solutions include injecting gases such as nitrogen into soles and eliminating laces altogether, with some designs pushing the total weight below the 100g threshold.
Adam Gilbertson, Managing Associate at Mathys & Squire, said: “We are also seeing footwear innovation become increasingly focused on comfort and motion support, as more people engage in running through communities and new trends that promote competition and performance monitoring. This is undoubtedly driving demand for technology that helps runners run faster and longer, and take on more demanding challenges like marathons with confidence.”
Nike is dominating the global trainer technology race, with 374 footwear patent applications published in 2025*, comfortably outpacing 18 other major competitors considered in this study (see graph below).
In fact, Nike had as many footwear technology patent applications published in 2025 as its five nearest competitors combined, which include Adidas, Sketchers, Asics, Puma and On. Nike’s long-time German rival, Adidas, increased its patent applications published by 88 in 2025, narrowing the gap but still trailing by a significant margin.
Among the patent applications by Nike is a “fluid movement controller” system embedded in a shoe’s sole, which enables users to adjust cushioning and support levels in real-time to adapt to running conditions and fatigue levels.
Other technologies aimed at improving runners’ performance for which patents are being pursued include:
- Shoe soles from On that return energy with every step. Flexible layers inside the sole stretch when you land and spring back as you push off.
- Adidas shoes that automatically shifts part of their sole sideways in response to foot pressure, to help keep the runner balanced.
- On’s running shoe midsole, which is designed to support runners by absorbing impact and smoothing uneven movement, helping running feel more stable and comfortable, even without perfect form.
- Adidas’ smart running shoes which have sensors that analyse movement and signals users to adjust their gait using vibrations or pressure cues.
- A running shoe that uses an internal air bladder with locking elements to adapt to the foot and then secure it in place, improving comfort and stability for long-distance runs.
Mathys & Squire says that while established footwear giants such as Nike, Adidas, Asics and Puma continue to innovate, data shows that newer rivals are developing their own technologies and investing more in patent protection. For example, over the past year, Swiss brand On had 49 patent applications published, breaking into the top 5 filers for the first time, while Lululemon had 11.
Rebecca Tew, Partner at Mathys & Squire, added that having invested in research and new technologies, footwear companies need to be wary of posting on social media too soon and ensure their intellectual property (IP) is protected via appropriate patent, design and/or trade mark filings. Otherwise, once displayed on social media, it may already be too late to seek patent protection for new technologies and innovations which might quickly be copied by competitors, making anti-counterfeiting particularly challenging.
The number of trainer patent applications rose 31% in the last year, with Nike leading the race
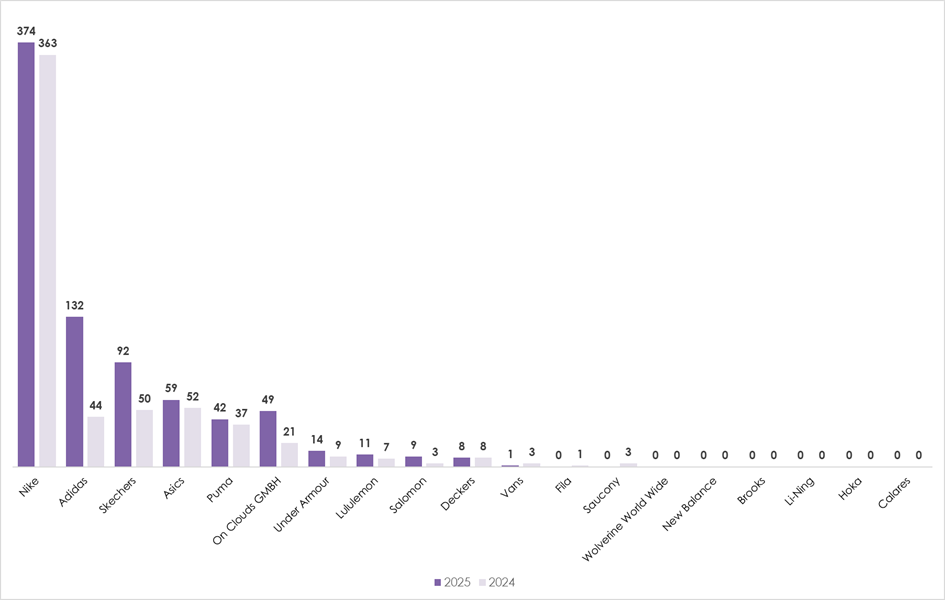
*Research conducted for calendar years 2025 and 2024 based on published patent applications.
In life sciences, the end goal of improving patient outcomes rarely changes. What has changed, quite dramatically, is the environment in which innovation must survive long enough to get there.
For early-stage companies in particular, the path from idea to impact is becoming harder to navigate. Capital is tighter, IPO windows have narrowed, and policy and macroeconomic conditions remain unpredictable. At the same time, global competition is intensifying, and the centres of innovation are shifting. Never has a robust intellectual property (“IP”) strategy been more critical.
A Market Under Pressure
Recent market activity illustrates this point. In the US, 2025 saw one of the weakest years for biotech IPOs in over a decade, with only nine companies listing. This is unlikely to be an isolated anomaly, but rather a reflection of a broader cooling in public market appetite for biotech, driven by weak sector performance, high interest rates, and a general shift away from risk.
This is only one side of the story. At the same time, the pharmaceutical sector is heading towards an unprecedented patent cliff, with more than $300 billion in drug revenues set to lose exclusivity between 2025 and 2030, as patents expire on nearly 200 products, including dozens of blockbusters. Companies therefore face rapid revenue erosion, often losing up to 80% of revenues within a year of generic or biosimilar entry. For large pharmaceutical companies, this creates immediate pressure to identify and secure new sources of value to mitigate against such a sharp drop.
The consequence is a market that feels constrained in one direction and highly active in another. While IPO activity has slowed, mergers and acquisitions and licensing activity remain strong. Transactions are happening, and at scale, but with a clear bias: large pharmaceutical companies are focusing on later-stage or near-commercial assets, where development risk is lower and the path to revenue is clearer.
A Divided Landscape
This has created a pronounced divide across the life sciences ecosystem. Later-stage biotech companies, particularly those with clinical or near-market assets, are in a relatively strong position. Demand remains strong, and competition between acquirers can drive attractive valuations.
Early-stage companies, by contrast, are operating in a much tougher funding environment. Traditional routes to capital are less reliable, and many are having to think earlier about partnerships, licensing strategies, or positioning for acquisition.
In this context, IP strategy becomes critical.
IP As the Core Asset
For an early-stage life sciences company, IP is what investors assess, what partners evaluate, and what acquirers ultimately buy.
A well-constructed patent portfolio can reduce perceived risk, create multiple pathways to monetisation, and support higher valuations. Conversely, a weak or poorly structured IP strategy can limit strategic options and undermine commercial outcomes.
Despite this, IP strategy is still too often treated as something that can be deferred to a later stage – in the current climate, that is a risk few companies can afford. Investing in an IP strategy early on (e.g. what to file, when to file, and how those filings evolve over time) can pay significant dividends for both commercial flexibility and exit opportunities.
Building an Effective IP Strategy
A company aiming for acquisition will structure its portfolio differently from one planning to commercialise independently. Likewise, a business expecting to rely heavily on licensing will need to think carefully about how clearly and broadly its core technology is protected, and how easily that protection can be understood and valued by third parties.
From there, execution becomes a matter of discipline and continuity. Early filings remain essential, particularly in fast-moving or competitive fields, but a single filing is rarely enough. Value is built over time through layered protection, follow-on patent applications that capture improvements, variations, and new uses, gradually strengthen the overall position and make it more difficult for competitors to design around.
There is also a more strategic dimension that is sometimes overlooked. If acquisition is a realistic outcome, it can be worth thinking in advance about who the likely acquirers might be. Understanding how your technology fits alongside their existing portfolios can inform your filing strategy in subtle but important ways. Patents that align with, extend, or reinforce an acquirer’s position can carry disproportionate weight in a transaction, leading to higher company valuations.
As companies mature, freedom to operate comes more to the fore. Demonstrating a clear understanding of the surrounding IP landscape, and the nature of competing rights, can significantly enhance credibility with investors and partners. In a more risk-averse funding environment, that kind of clarity is increasingly valuable.
A Shifting Global Landscape
Alongside these strategic considerations, the geographic dimension of IP is evolving. For many years, the default approach in life sciences was to focus heavily on the US and Europe. Those jurisdictions remain central, but the balance is shifting.
In the US, there are signs of strain within the patent system. Operational disruption at the United States Patent and Trademark Office combined with changes to examination processes is creating greater uncertainty around timelines to grant. For companies reliant on patent protection to support investment and partnering, this unpredictability can have tangible commercial consequences.
Europe, by contrast, has remained relatively stable. The European Patent Office continues to process a high proportion of cases in line with targets, and in many instances offers a more predictable route to grant. That reliability can be valuable when coordinating multi-jurisdictional strategies.
Perhaps the most significant change is the continued rise of Asia, particularly China. Patent filing activity in this region has grown rapidly, far outpacing historical trends in other regions. This is not just a numbers story – it reflects a broader shift in where innovation is happening and how it is being commercialised.
The Rise of Cross-Border Licensing
A result of this shift is the increasing importance of cross-border licensing. As supply chains become more uncertain, affected by tariffs, policy changes, and geopolitical tensions, companies increasingly look for ways to reduce reliance on the physical movement of goods. Licensing allows businesses to access and commercialise innovation across jurisdictions without relying on physical product movement.
This approach brings its own complexities, particularly in relation to manufacturing, tax and structuring considerations. However, it also offers increased flexibility and can enable more efficient global deployment of innovation while retaining greater control over supply chains and cost structures.
The growth of outbound licensing from China is a clear example of this trend. Deal volumes and values have increased significantly, particularly in high-value therapeutic areas such as oncology and advanced biologics. Notably, many of these transactions involve relatively early-stage assets that combine strong scientific differentiation with clear commercial potential.
Standing Out in a More Competitive Market
For UK-based life sciences companies, the implications are clear. Competition is becoming more global, more strategic, and more selective.
Strong science remains essential, but it is not sufficient on its own. Companies must be able to demonstrate a clear and coherent approach to intellectual property – one that supports their commercial strategy, aligns with market expectations, and resonates with investors and acquirers.
That means thinking early, acting deliberately, and building a portfolio that not only protects the core innovation but also tells a compelling commercial story.
In today’s market, patents are not just defensive tools, they are strategic assets that can unlock funding, enable partnerships, and ultimately determine whether an innovation reaches the patients it is intended to serve.
Contact our team
Reach out to Mohammed Shafi and Craig Titmus from our Life Sciences & Chemistry team today to help understand the value of your assets and build your own IP strategy.