The Supreme Court judgment (24 June 2020) sends a clear “no” Brexit message to any big pharma contemplating corporate muscle-flexing of excessively broad patent claims. This ruling overturned the position held by the Court of Appeal that, for patents relating to “a principle of general application”, there was no requirement to teach how to make the full range of claimed products. In this regard, the Court of Appeal held that Regeneron’s contribution to the field extended beyond the products (transgenic mice) that could be made back in 2001, and instead related to the general principle of providing ‘better’ mice (thereby overcoming a prior art immuno-sickness problem inherent to mice transfected with human DNA). With hindsight, the Court of Appeal allowed too much weight to be given to post-invention evidence of success submitted by Regeneron, and to the relative contribution the ‘better’ mice aspect provided in producing a(ny) mouse having commercial utility. In sum, the Supreme Court considered the Court of Appeal had incorrectly watered down the “sufficiency of disclosure” requirement of patent law and, in doing so, this judgment maintains a sensible balance between patent law enforceability and invalidity.
Background to the case
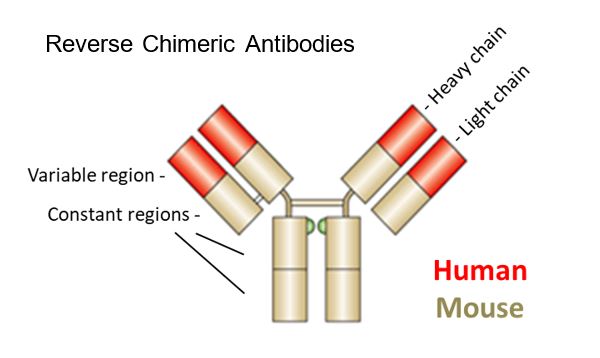
Regeneron obtained two patents with a priority date of 16 February 2001, EP(UK) 1 360 287 (“the 287 Patent”) and EP (UK) 2 264 163 (“the 163 Patent”, a divisional of the 287 Patent). At the priority date there were two main problems associated with the use of mice as a platform for antibody development. First, use of murine antibodies in humans typically resulted in a host rejection response. Secondly, the transfection of mice with human antibody genes was associated with the mice developing a reduced immune response function (and thus reduced antibody titres). Regeneron’s solution to this reduced antibody production capability was to develop a hybrid (chimeric) antibody gene structure, consisting in part of human and in part of murine elements, created by insertion of ‘human variable regions’ into the genome of the mouse, whilst retaining the ‘mouse constant regions’. It is, of course, the ‘variable’ regions that are primarily responsible for antibody recognition of its target antigen, and thus essential for generating antibodies against new targets.
Kymab’s challenge to validity arose in defence to an infringement action brought by Regeneron against Kymab’s commercialisation of its own transgenic mice, “Kymouse”. At first instance (Patents Court), Mr Justice Henry Carr [[2016] EWHC 87 (Pat)] found that the difficulties in producing a hybrid gene structure where the whole of the human variable region was combined with the murine constant region were not taught (enabled) by the technical disclosures offered by the Regeneron patents (even when combined with the common general knowledge at the time of the priority filing), concluding that “at the priority date, the skilled person would not have been able to perform the invention over the whole area claimed without undue burden and without needing inventive skill”. This decision was overturned by the Court of Appeal [[2018] EWCA Civ 671], holding that Regeneron’s patents contained enough information to insert some of the human material into a mouse’s genes. Whilst this would have created a hybrid mouse (as claimed), the prevailing genetic manipulation techniques available imposed a significant limitation on the amount of human DNA that could be transferred. Indeed, said limitations would, at best, have permitted the transfer of up to 5% of the human variable region V segments into a mouse genome. Moreover, the necessary transfection techniques required for transfer of the full human repertoire were not invented until 2011. Thus, the Regeneron patents taught how to make a transgenic mouse having up to 5% of the necessary human repertoire, which as such simply had no meaningful commercial utility. Despite this, however, the Court of Appeal upheld the Regeneron patents on the basis they related to a “principle of general application”, and did not therefore require any teaching of how to make products commensurate with the scope of the claims. Kymab appealed to the Supreme Court.
The claim in question covers a range of mice
The case turned on the relevance or otherwise, of the existence of a very narrow range of mice having amounts (up to 5%) of the human variable domain repertoire, to the question of sufficiency. The appellant submitted that the range was of the highest importance because of its effect upon the ability of a particular type of mouse to produce a wide variety of B cells, and hence its potential to deliver a broad stream of useful antibodies (and thus have an meaningful commercial utility). The respondent/patentee submitted that the existence of this range was irrelevant, because the unique advantage conferred by the use of a Reverse Chimeric Locus, namely a cure for the immunological sickness of the recipient mouse, worked across the whole range, regardless of the amount of the human variable region DNA inserted into the murine genome, because it retained the murine constant region genes.
Lord Briggs reviewed of sufficiency in the UK and EPO
In considering UK cases (Biogen Inc v Medeva plc [1995] RPC 25 and [1997] RPC 1, Kirin-Amgen Inc v Hoechst Marion Roussel Ltd [2005] RPC 9 Generics (UK) Ltd v H Lundbeck A/S [2008]; Lord Briggs found that the UK case law provides clear guidance for product claims, in contrast to the situation for a process such as described (EPO case law) in ‘Polypeptides’ (Genentech I/Polypeptide expression (1988) (T 292/85) 1988), reciting Hoffmann on insufficient disclosure: “The patent may claim results which it does not enable, such as making a wide class of products when it enables only one of those products and discloses no principle which would enable others to be made.” (Biogen) “In the case of a product claim, performing the invention for the purposes of section 72(1)(c) means making or otherwise obtaining the product. In the case of a process claim, it means working the process. A product claim is therefore sufficiently enabled if the specification discloses how to make it” (emphasis added).
Lord Briggs set out principles for determining sufficiency
i) The following principles for determining sufficiency were outlined [56]:The requirement of sufficiency imposed by Article 83 of the EPC exists to ensure that the extent of the monopoly conferred by the patent corresponds with the extent of the contribution which it makes to the art.
ii) In the case of a product claim, the contribution to the art is the ability of the skilled person to make the product itself, rather than (if different) the invention.
iii) Patentees are free to choose how widely to frame the range of products for which they claim protection. But they need to ensure that they make no broader claim than is enabled by their disclosure.
iv) The disclosure required of the patentee is such as will, coupled with the common general knowledge existing as at the priority date, be sufficient to enable the skilled person to make substantially all the types or embodiments of products within the scope of the claim. That is what, in the context of a product claim, enablement means.
v) A claim which seeks to protect products which cannot be made by the skilled person using the disclosure in the patent will, subject to de minimis or wholly irrelevant exceptions, be bound to exceed the contribution to the art made by the patent, measured as it must be at the priority date.
vi) This does not mean that the patentee has to demonstrate in the disclosure that every embodiment within the scope of the claim has been tried, tested and proved to have been enabled to be made. Patentees may rely, if they can, upon a principle of general application if it would appear reasonably likely to enable the whole range of products within the scope of the claim to be made. But they take the risk, if challenged, that the supposed general principle will be proved at trial not in fact to enable a significant, relevant, part of the claimed range to be made, as at the priority date.
vii) Nor will a claim which in substance passes the sufficiency test be defeated by dividing the product claim into a range denominated by some wholly irrelevant factor, such as the length of a mouse’s tail. The requirement to show enablement across the whole scope of the claim applies only across a relevant range. Put broadly, the range will be relevant if it is denominated by reference to a variable which significantly affects the value or utility of the product in achieving the purpose for which it is to be made.
viii) Enablement across the scope of a product claim is not established merely by showing that all products within the relevant range will, if and when they can be made, deliver the same general benefit intended to be generated by the invention, regardless how valuable and ground-breaking that invention may prove to be.
Application of those principles to the current case shows that Claim 1 fails for insufficiency, because only a small range of the products could be made, and mice at the more valuable end of the range could not be made.
Interesting observations (unusual nature of the Court of Appeal Order)
Kymab obtained a stay of injunction, order for delivery up and disclosure granted pending an application to appeal to the Supreme Court in respect of Kymab’s collaborative work with humanitarian bodies including the Bill & Melinda Gates Foundation and the International AIDS Vaccine Initiative to treat diseases with unmet clinical need. Under the terms of the stay, Kymab was permitted to ‘dispose or export’ antibodies or mouse serum for the purposes of:
Kymab’s collaborations with the Bill & Melinda Gates Foundation and the International AIDS Vaccine Initiative and Heptares Therapeutics Limited; and
For preparing and conducting pre-clinical or clinical trials, antibody producing CHO cells for use by Kymab’s manufacturing CRO Lonza (and which will remain under Kymab’s control) solely for the purposes of manufacturing antibodies under GMP conditions for use in pre-clinical or clinical trials.
Summary
A patent reflects a bargain between the inventor and the public. The inventor gains a time-limited monopoly over the making and use of a product. In return, the public gains the ability to make the product after the expiry of the monopoly. As part of this bargain, the inventor must publish sufficient information to enable a skilled member of the public to make the product. This ensures that patent holders only gain legal protection which is proportional to their actual technical contribution to the art, and encourages inventors to conduct research for the benefit of society. The decision makes a clear distinction between the requirements for a product claim (that the product can be made across the breadth of the claimed range at the effective date of the patent), and a process claim, where the process can be applied to a range of inputs, and will always provide the benefit. Whereas the Court of Appeal gave the patentee a tantalising glimpse of a world where the patentee might receive the benefit for having a good idea, even though they couldn’t make it work across the board, this decision puts the balance back firmly with the public.



